Thirona, a company specializing in artificial intelligence (AI) for medical image analysis, has obtained an exclusive license on a patent in relation to Cystic Fibrosis (CF), a rare genetic condition that creates lung abnormalities from a very early age. The patent allows Thirona to develop an AI algorithm that will analyze CT scans to identify even the smallest abnormalities in the lungs, especially early on, ensuring that the right treatment can follow. The AI software is expected to have a substantial impact on patients suffering from CF, as it paves the way for earlier and more personalized treatment of the disease, especially for cystic fibrosis in children.
Building on Existing Research
Worldwide, there are approximately 70,000 known cases of CF, making it a rare genetic disorder. Years of research on the disease, conducted by the Erasmus University Medical Centre Rotterdam in the Netherlands and the Telethon Kids Institute in Australia, previously resulted in PRAGMA-CF: a quantitative method for the analysis of chest CT scans of children with CF. The current PRAGMA-CF method allows clinicians to manually assess the extent of lung disease in children with CF. However, the use of PRAGMA-CF requires extensive training and is time-consuming.
Medical artificial intelligence company Thirona, which has worked on diseases like COPD, Asthma, and Tuberculosis, has now obtained an exclusive license to a patent to integrate the method into its certified LungQ software, as well as co-exclusive rights to use the underlying datasets of PRAGMA-CF. By leveraging artificial intelligence, clinicians will have an automated method to detect and quantify the extent of CF lung disease – thus helping to improve care and clinical decision-making for these patients.
Artificial Intelligence for Cystic Fibrosis
“Chest CT is the best way to assess the structural changes related to CF lung disease even at an early stage in young children. PRAGMA-CF, developed and validated together with the Telethon Kids Institute, provides a sensitive and quantitative image analysis method to assess the extent of CF lung disease,” says Prof. Harm Tiddens, MD, PhD, from Erasmus MC. “The collaboration with Thirona is of key importance for the development of fully automated PRAGMA-CF software. Their scientists have not only artificial intelligence expertise but also in-depth knowledge of the lung. This allows them to ask us the right questions needed for the development of sensitive and accurate software. Integration of PRAGMA-CF into their LungQ platform will make it accessible for CF clinicians around the world for clinical care as well as for clinical studies.”
Proud to Make a Difference
“Artificial intelligence algorithms make medical image analysis easier, more accurate, and more effective. But it is hard work,” says Prof. Eva van Rikxoort, PhD, managing director of Thirona. “Developing an algorithm that works as well as – and sometimes even better than – a clinician, takes time. This is especially the case when developing a solution/algorithm to be used on the analysis of CT scans of children. They are still growing, and their bodies and organs therefore keep changing, with or without a condition. All these changes with age must be calculated into the algorithm; a time-consuming process, but one that is worthwhile. The algorithm we are developing will be able to quickly and accurately detect bronchiectasis and mucous quantifications in very young patients with CF, making personalized treatment easier. We believe this will have a substantial impact for patients and are proud to be part of the process.”
For more information visit https://thirona.eu.
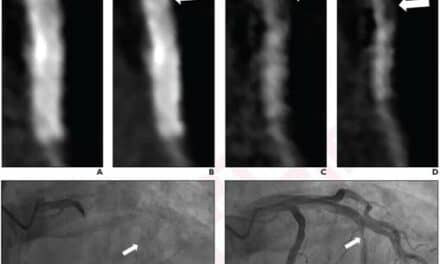
Featured image: CT scans of (A) a patient with normal lungs and (B) a patient with severe cystic fibrosis, analyzed with PRAGMA-CF. This analysis quantifies disease patterns related to CF, including normal lung tissue (green), bronchiectasis (blue), airway thickening (red) and mucus plugging (pink). Thirona is developing an automated AI solution to quickly and accurately quantify PRAGMA-CF disease patterns. (Credit: Thirona)