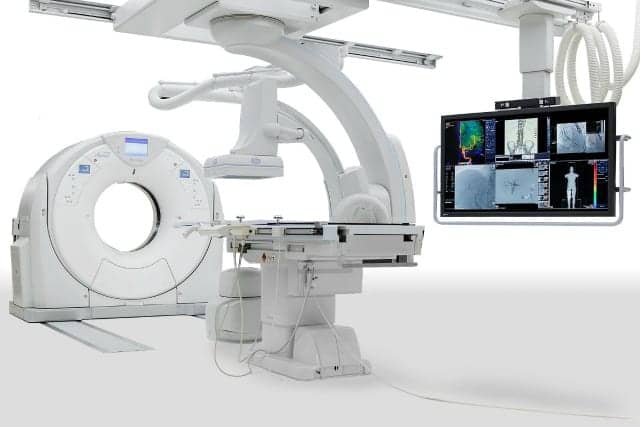
Toshiba America Medical Systems Inc., of Tustin, Calif, recently announced FDA approval of its new Infinix 4DCT with the Aquilion PRIME CT system configuration. Providing features and tools for clinicians to optimize their time, the company aims to improve the patient experience by bringing CT technology to the interventional lab.
Toshiba America Medical Systems Inc., of Tustin, Calif, recently announced FDA approval of its new Infinix 4DCT with the Aquilion PRIME CT system configuration. Providing features and tools for clinicians to optimize their time, the company aims to improve the patient experience by bringing CT technology to the interventional lab.
The Infinix 4DCT previously received FDA clearance with the Infinix Elitecardiovascular X-ray system and Aquilion ONE ViSION Edition CT system configuration.
As an all-in-one interventional lab and CT solution to deliver real-time CT images during procedures, Infinix 4DCT features SUREGuidance technology, which allows for automatic transition between modalities. It is also designed to save significant time by allowing clinicians to perform CT and interventional procedures within the same room and verify treatment success following procedures. The solution streamlines the workflow of oncology and cardiac procedures, providing interventionalists with CT images for the target anatomy.
“The Infinix 4DCT allows clinicians to plan, treat and verify in a single clinical setting, which negates the need to transfer patients between departments, resulting in anticipated decreases of both procedure time and risk of infection,” said Bill Newsom, director, X-ray vascular business unit. “This also contributes to improved patient safety and satisfaction by minimizing the clinicians’ time spent away from patients, allowing them to focus on providing quality care.”
Get AXIS e-newsletters free. Subscribe here.