|
· Philips and Celsion Partner on Drug-Delivery Research
· Portable Ultrasound Combines Quality, Convenience
Philips and Celsion Partner on Drug-Delivery Research
What does one get when combining MR-guided high-intensity focused ultrasound (HIFU) with an anti-cancer-fighting drug encased in a temperature-sensitive liposome? Netherlands-based Royal Philips Electronics and Celsion Corp, Columbia, Md, are expecting a new cancer treatment that will safely and effectively target tumors.

|
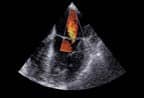
| MR units, such as Philips’ Achieva series, can be used to track the drug and guide the HIFU in drug delivery systems. |
The two companies signed a multiyear joint research agreement to explore the combined use of Philips’ investigational MR-guided HIFU and Celsion’s leading drug candidate, ThermoDox, to treat a broad range of cancers. The researchers will use the imaging modalities to position the drug and release it by creating a mild hyperthermia.
The team has two theories to test. The first is to determine whether ThermoDox has the ability, when delivered and released via MR-guided HIFU, to kill the cancer cells that lie around the heated area’s margins and are responsible for secondary tumors. The second theory is to explore the potential for the two technologies to deliver high concentrations of chemotherapeutics to multiple sites anywhere within the body, including those areas previously inaccessible.
“Localized drug delivery is being evaluated to determine the potential for increasing the potency of drugs in their target regions with equal or less toxicity to critical organs,” said Falko Busse, vice president and chief technology officer of MR for Philips Healthcare, in a press release.
MR-guided HIFU can improve the accuracy of treatment, increasing the intensity on a targeted lesion while limiting damage to surrounding tissue. MR is used to track the drug (which is enhanced with contrast); HIFU thermally activates it once the medication is at the treatment site.
According to the Web site of Philips Applied Technologies, Atlanta, the company is developing a removable HIFU add-on for existing MR systems. One prototype has been designed to integrate with the patient table; Philips Applied Technologies has contributed the actuating mechatronics for steering the ultrasonic probe within the table as well as the steering software.
Volumetric heating with feedback technology will allow control of both the tissue temperature and the heating area. MR measurement techniques help to ensure a specific temperature.
“Philips’ MRI-guided HIFU technology has the potential to heat a defined area while providing a real-time visualization of the heated area through the use of MRI,” Michael H. Tardugno, Celsion’s president and CEO, said.
ThermoDox releases doxorubicin, a proven anticancer agent, at a target temperature of 39?C to 42?C. Until that point, the drug is encapsulated within a lysolipid thermally sensitive liposome.
The company has a clinical trial in progress examining the use of radiofrequency ablation (RFA) with ThermoDox in the treatment of hepatocellular carcinoma. Another study is examining the use of ThermoDox in conjunction with microwave heat to treat advanced breast cancer patients with loco-regional chest wall disease.
The goal of this research as well as that using MR-guided HIFU is to intensify the delivery of the anticancer medication to the lesion while reducing the exposure of nearby normal tissue to the drug and the associated side effects. Previously inaccessible tumors may now become accessible.
Philips and Celsion will look at a number of solid tumor cancers, working with those that may be susceptible to a high concentration of doxorubicin and concurrent hyperthermia. Tardugno has high hopes for the research. “If our thesis proves to be correct, the Celsion-Philips tumor-targeting combination has the potential to shift the paradigm for the treatment of primary and metastatic disease.”
—R. Diiulio
Portable Ultrasound Combines Quality, Convenience
The fundamentals for portable ultrasound include not only those characteristics conducive to mobility, but also those qualities required in a clinically useful imaging system, according to Dave Willis, vice president of innovation and competitive strategy for SonoSite Inc, Bothell, Wash. The company has launched its SonoGT Global Targeted technology, designed to meet both of these needs while maximizing the company’s most advanced portable ultrasound platform, the M-Turbo.

|
| In this ultrasound image, the M-Turbo with SonoGT captures carotid artery plaque. |
“We feel we’ve approached the image quality and color sensitivity that [users of portable ultrasound need] but also kept the system durable, portable, and easy to use for physicians doing the scanning. We never want to give up on those things that are essential for point-of-care ultrasound,” Willis said.
What the company does want to achieve is the development of products that meet the needs of its customers. “Everything we tend to add in a build is generally based on watching customers do a procedure and trying to make it better or faster for them or the patient,” Willis said.
Subsequently, the SonoGT Global Targeted technology includes advances intended to increase the image capabilities and clinical productivity available in a portable system. The platform incorporates a new level of color flow imaging, wireless connectivity, and workflow integration for anesthesia, emergency medicine, critical care, and other acute point-of-care markets.
All of the advances contribute to faster speeds, which can improve the patient experience, allow increases in volume, grow revenue, and save money. “If you shave 20 to 25 minutes from a surgical procedure and look at the cost of the time in the surgical suite, shaving off 20 minutes has a huge dollar value attached to it,” Willis said.
Those 20 minutes can be gleaned from simple changes. A new accessory debuting at the same time as the SonoGT is expected to introduce cost savings through greater productivity and reduced staff during procedures. The SonoRemote Control is exactly what it sounds like—a remote control for the ultrasound unit.
The device is wireless, voice activated, and sterilizable, meaning it can operate within a sterile bag or sheath. “Quite often users are in a sterile environment, wearing gloves and unable to touch the machine to make modifications. Touch screen technology doesn’t really help because when gloved up, the physician can’t touch anything and has to ask a nurse or assistant, which can be very cumbersome,” Willis said.
The SonoRemote Control can activate the ultrasound system by the physician’s voice or touch and works within a radius of 10 meters. An assistant is no longer needed to work the machine, Willis notes. In addition, the ultrasound machine can be placed on the other side of the patient bed from the clinician without worrying about proximity or the amount of other equipment in the room.
Another time-saving new feature of the SonoGT is the ability to complete data transmission wirelessly. Using SonoRoam Technology, images are sent from the M-Turbo system to a PACS system via DICOM or to a personal computer via SonoSite’s SiteLink.
Transmission can occur while the user is mobile, moving from one patient to the next. Willis explains that in conventional imaging, the sonographer would take the device to the patient’s department, where he or she would complete the scan, and then return to radiology to download the images. On a busy morning, that return trip could be delayed until all patients had been scanned.
“In some cases, the physician reading the cases had to wait a half or full day to even receive the images to read,” Willis said. Wireless transfer means the images can be delivered and read immediately.
The images are also easier to interpret thanks to the company’s new ColorHD Technology. The proprietary, color Doppler algorithm increases color performance, sensitivity, and frame rates. It also comes with a variable sector size for the P10x/8-4 MHz and the P21x/5-1 MHz phased array transducers for increased frame rates during cardiac imaging. “[ColorHD] represents a big color improvement, and we can now visualize very small blood vessels with low-velocity blood flow,” Willis said.
Other system improvements also impact workflow. The addition of DICOM capabilities facilitates seamless clinical integration of ordering, scheduling, image acquisition, storage, viewing, and billing of patient procedures. A new USB barcode reader enables clinicians to enter patient information and update exam information prior to its entry into the patient record, a benefit that is especially valuable in the emergency department where the health care team often wants to start patient diagnosis and treatment as quickly as possible.
“We don’t want to replace the 600-pound machine, but our technology provides a portable solution for studies that need to take place in the various wards,” Willis said. SonoSite will begin shipping the new M-Turbo systems with SonoGT technology this quarter and the SonoRemote Control in the first quarter of 2009. Customers with existing M-Turbo systems have the option of upgrading via an easy software download from a USB thumb drive.
—Renee Diiulio




